Flow cytometry for the study of hematologic neoplasms
By: Dr. Vanessa Santiago Pacheco
Medical Pathologist, Laboratorio Clínico Hematológico S.A. Medellín, Colombia.
Published on 23-07-2021
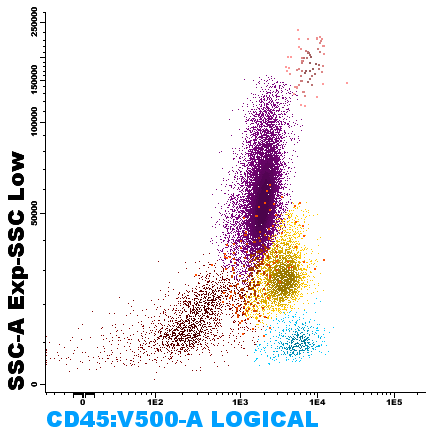
Figure 1. Visualization of cell populations in marrow blood, according to their cytoplasmic complexity (SSC-A) and CD45 (Leukocyte Common Antigen) expression.
Flow cytometry is a rapid, reliable and inexpensive tool to establish the monotypic or polytypic origin of a cell population, which allows its use to assess bone marrow involvement by lymphomas and other hematopoietic diseases
Immunophenotyping by flow cytometry is an essential component in the diagnosis of acute leukemias; it allows rapid and comprehensive assessment of the relevant membrane and intracellular antigens expressed by leukemic cells according to their maturation stage and progeny, which facilitates the identification and characterization of blasts, cell lineage assignment, assessment of possible therapeutic targets and prediction of some genetic alterations.
Peripheral and medullary blood are the samples most commonly analyzed by flow cytometry; however, any tissue in which individual cells can be isolated in suspension can be analyzed in this way, e.g., cerebrospinal, pleural and ascitic fluid, even lysed samples of solid tissues such as lymph nodes.
Applications:
Flow cytometry is a laboratory test, with rapid processing, high analytical sensitivity and specificity, that allows the characterization of cell populations in a sample; with applications in the accurate and timely diagnosis, classification, prognosis and treatment monitoring of numerous hematologic malignancies. This technology simultaneously measures and analyzes multiple characteristics of cells as they are suspended in a system fluid and pass through a beam of light, such as relative size, relative complexity or granularity, and relative fluorescence intensity. These characteristics are determined using an optical-electronic system that records how the particle (or cell) scatters light or emits fluorescence.
This method of study has been employed for more than 40 years in the diagnosis and monitoring of hematopoietic neoplasms, through the use of fluorochrome-labeled monoclonal antibodies, which are each directed against a specific molecule on the surface, cytoplasm or nucleus of leukocytes and hematopoietic precursors. Initial studies were performed with single antibodies or with a combination of two antibodies, which limited the sensitivity for detecting minority cell populations. With the availability of new fluorochromes and stains, the current analysis is routinely performed with 8 to 12 different antibodies in a single sample tube.
Advantages and limitations:
Flow cytometry has proven to be a very useful and versatile tool in the diagnosis and monitoring of hematological diseases, in addition to numerous other applications. Advances in electronics, software and reagents in recent years have simplified some aspects of flow cytometry and made it possible to combine different antibodies in a single sample tube, which in turn has facilitated the identification of abnormal populations. This is a very sensitive and objective technique that allows the analysis of a large number of cells in a short time (5,000 to 20,000 events/second) and the use of more than one antibody at a time to identify subpopulations within a heterogeneous population. This ability to acquire large numbers of cells (events) has made it possible to detect low numbers of abnormal cells, which may persist after treatment, to measure minimal residual disease and to add these data to personalized treatments.
Historically, flow cytometry has been perceived as a technique highly dependent on experience and with low reproducibility; however, the development of consensus has allowed the development of standardized panels for rapid and accurate diagnostic approach, in addition to the classification of hematological neoplasms and their follow-up, such as the Euroflow® consortium 10. Another limitation that has been described in the study of flow cytometry is the absence of a morphological assessment of neoplastic cells, since this is an essential part of the diagnosis and classification of hematopoietic diseases. This is why immunophenotype assessment should be performed in conjunction with observation and analysis of the biopsy of the affected tissue, such as peripheral blood smear, bone marrow biopsy and aspirate, or lymph node biopsy. In the ideal context for the patient, these samples should be evaluated by the same specialist to arrive at a comprehensive diagnosis.
The Laboratorio Clínico Hematológico in the classification of hematological malignancies:
We have flow cytometry for bone marrow samples, peripheral blood, liquids and tissue biopsies, adjusted to the protocols and panels determined by the Euroflow®consortium and with access to its databases to achieve a reproducible diagnosis. Our turnaround time for results is two working days, with immediate telephone reporting in case of obtaining critical values, such as the diagnosis of acute leukemias or high-grade lymphomas. From the Diagnostic Hematology Division, we offer an Integrated Hematopathology Report, both for our own cases and for the specialized interconsultation service; a report that, beyond the report of histopathological and immunophenotypic findings, provides a complete and specialized analysis of each patient in a global context. This, in order to provide a clinical, morphological and laboratory correlation that allows the treating physician a practical approach that adds value to the management and clinical benefit of the patient.
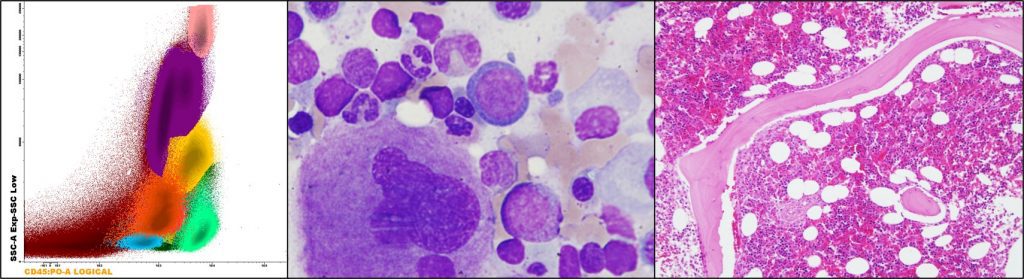
Figure 2. Cellular populations in bone marrow, from left to right: flow cytometry, aspirate, and biopsy.
Bibliography
- BD Biociencias. Introducción a la citometría de flujo.pdf. BD Biociencias; 2005.
- Barlogie B, Latreille J, Freireich EJ, Fu CT, Mellard D, Meistrich M, et al. Characterization of hematologic malignancies by flow cytometry. Blood Cells. 1980;6(4):719-44.
- Bernstein ID, Andrews RG, Cohen SF, McMaster BE. Normal and malignant human myelocytic and monocytic cells identified by monoclonal antibodies. J Immunol Baltim Md 1950. febrero de 1982;128(2):876-81.
- van Dongen JJM, Lhermitte L, Böttcher S, Almeida J, van der Velden VHJ, Flores-Montero J, et al. EuroFlow antibody panels for standardized n-dimensional flow cytometric immunophenotyping of normal, reactive and malignant leukocytes. Leukemia. septiembre de 2012;26(9):1908-75.
- Hedley BD, Keeney M, Popma J, Chin-Yee I. Novel lymphocyte screening tube using dried monoclonal antibody reagents. Cytometry B Clin Cytom. diciembre de 2015;88(6):361-70.
- Rajab A, Axler O, Leung J, Wozniak M, Porwit A. Ten-color 15-antibody flow cytometry panel for immunophenotyping of lymphocyte population. Int J Lab Hematol. mayo de 2017;39 Suppl 1:76-85.
- Sorigue M, Cañamero E, Miljkovic MD. Systematic review of staging bone marrow involvement in B cell lymphoma by flow cytometry. Blood Rev. mayo de 2021;47:100778.
- DiGiuseppe JA, Wood BL. Applications of Flow Cytometric Immunophenotyping in the Diagnosis and Posttreatment Monitoring of B and T Lymphoblastic Leukemia/Lymphoma. Cytometry B Clin Cytom. julio de 2019;96(4):256-65.
- Cherian S, Hedley BD, Keeney M. Common flow cytometry pitfalls in diagnostic hematopathology. Cytometry B Clin Cytom. noviembre de 2019;96(6):449-63.
- van Dongen JJM, Orfao A, EuroFlow Consortium. EuroFlow: Resetting leukemia and lymphoma immunophenotyping. Basis for companion diagnostics and personalized medicine. Leukemia. septiembre de 2012;26(9):1899-907.



